The European Union (EU) has started developing a “fax vax” in preparation for the “next pandemic.”
The German-EU “Vaccelerate” program is already on the lookout for the next “pathogen with pandemic potential.”
Scientists affiliated with the program have detailed the plan in a new paper titled “Predicting the next pandemic.”
The paper is devoted to a survey of the Vaccelerate consortium’s own members on the pathogen most likely to produce the “next pandemic” – or the next “potential pandemic generator.”
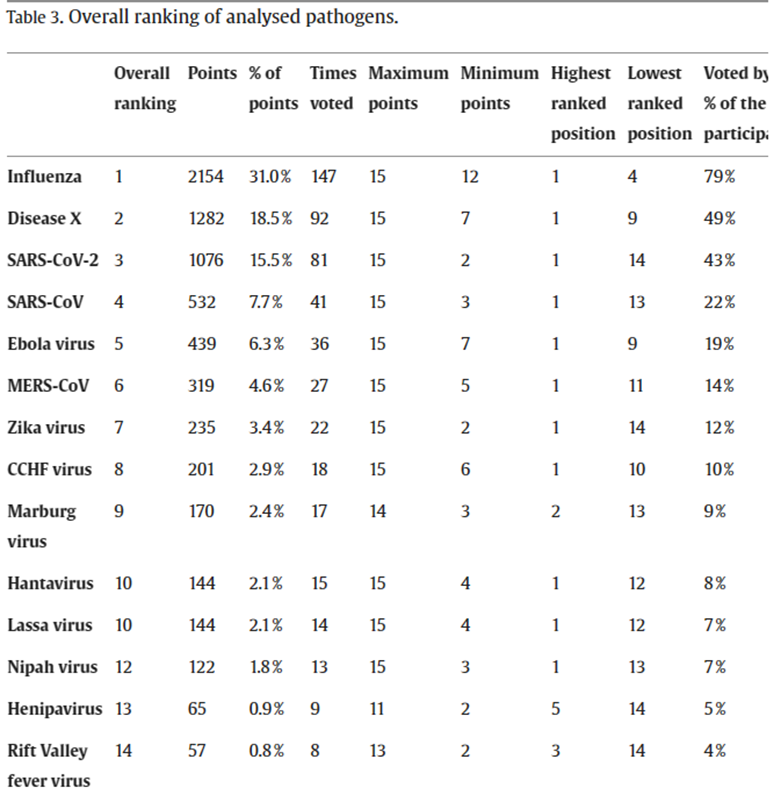
The responses involved a ranking of the various candidates and were tallied using a points system.
Influenza was named by nearly 80% of respondents and garnered nearly one-third of the points.
A hitherto unknown “Disease X” finished second, and the Coronaviruses, SARS-CoV-2 and SAR-CoV-1, finished third and fourth respectively.
The full results are reproduced below.
The Vaccelerate program grew out of the European Vaccine Trial Accelerator Platform (EUVAP), which was founded with a €600,000 grant from the German Government at the University of Cologne in 2020, during the initial rush to create a COVID-19 vaccine. (See here under “Vaccelerate”.)
In early 2021, Vaccelerate as such was created as an EU-wide network with a major grant from the European Commission.
Per its EU funding page here, the Vaccelerate program has received no less than €26.5 million in EU funding overall.
According to the project description on the funding page, the program’s original purpose, in keeping with the objective of EUVAP, was to create an EU-wide network of clinical trial sites and a registry of willing trial participants in order to fast-track specifically COVID-19 vaccine candidates.
“The ongoing COVID-19 pandemic creates an unprecedented burden worldwide” the description reads:
Vaccine-induced immunity is the only promising solution.
There is continued need for phase two and three vaccine trials to reach long-term, large-scale immunity of the entire European population.
Vaccelerate will be the pan-European backbone accelerating phase two and three COVID-19 vaccine trials.
With COVID-19 vaccines having already been rolled out in record time shortly before its launch, by the time EUVAP had morphed into Vaccelerate in 2021.
However, this original purpose was, of course, largely obsolete.
Hence, the program’s pivot toward the next “pathogen with pandemic potential” is hardly surprising.
Indeed, the original project description already notes that “Beyond the COVID-19 pandemic, [Vaccelerate] will be an established pandemic preparedness network, ready to face emerging future pandemics” and “enhance vaccine development capacity in Europe”.
The Vaccelerate website notes, moreover, that the program “is funded by the European Commission’s activities for future pandemic preparedness, the HERA Incubator.”
HERA is the EU’s Health Emergency Preparedness and Response Authority, which was likewise created in 2021.
The Vaccelerate consortium is led by the Clinical Trial Unit of the German Centre for Infection Research (DZIF). The Clinical Trial Unit is based at the University of Cologne.
The DZIF is a German public agency that partners with pharmaceutical companies in developing vaccines.
One of the DZIF’s partners is none other than BioNTech.
See the screenshot from the DZIF website below.
BioNTech is the German developer and in fact legal manufacturer of what is more commonly known as the ‘Pfizer’ vaccine.
Pfizer performs (some) manufacturing activities as a contract manufacturer on BioNTech’s behalf (see here).
BioNTech has also earned far more than Pfizer on sales of the drug.

The head of the DZIF’s Product Development Unit is none other than Klaus Cichutek, who is at the same time the President of the German vaccine regulator, the PEI or Paul Ehrlich Institute (so-named for the German immunologist, not the American population control theorist).
It is this dual role of enabler and regulator that raises obvious questions about the impartiality of the PEI’s oversight of the BioNTech vaccine, and these questions are all the thornier given the leading role which, by Cichutek’s own admission, the PEI plays at the European Medicines Agency (EMA).
Oliver Cornely of the University of Cologne is both the Vaccelerate project leader and the coordinator of the DZIF’s Clinical Trial Unit.

Yeah,
smart people didn’t take the last 1, so do they think we would get STUPID or something???
I vote for the Zika Virus as I have been seeing gazillion of 5-year-old pinheads walking the streets lately. Just needs a little more gain-of-function tweaking. /s
The covid-19 vaccine has destroyed the public’s confidence in vaccines. It may take a generation to rebuilt the public’s confidence is MRNA technology and any vaccines regardless of how they are created. The response of course will be to make taking the vaccine legally mandatory which will make more people to decide against taking another shot. Time to let this cool down and let people recover from the last failed attempt to cause widespread death in the name of healthcare.
THAT PARASITE GATES AND HIS PALS CAN SHOVE THAT STRAIGHT UP THEIR PATHETIC ASSES!!! WHO THESE SCUM THINK THEY ARE???
Evidently the DZIF didn’t get the “memo” from the previous article
F*ck them and F*ck their so called vaccine!